Cosmetic Safety and Compliance: Stop Worrying About Regulatory Risks
The Regulatory Nightmare
Launching a beauty brand should feel exciting. Instead, many founders lie awake at night worrying about regulatory compliance.
Will my product pass safety testing?
Am I using banned ingredients?
Does my labeling meet FDA requirements?
What if I get hit with a recall?
Can I sell in Europe with these formulations?
These fears are valid. Regulatory violations can destroy brands. Fines, recalls, and legal action cost far more than prevention. But here is what most founders do not realize: compliance does not have to be complicated, expensive, or stressful.
The key is shifting from reactive panic to proactive systems. This guide shows you how.
Why Compliance Feels Overwhelming
The Rule Maze
Cosmetic regulations vary dramatically by market:
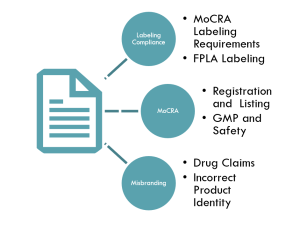
United States: FDA oversight under MoCRA (Modernization of Cosmetics Regulation Act), which now requires facility registration, product listing, and serious adverse event reporting
European Union: Strict EU Cosmetics Regulation with banned ingredient lists, safety assessments, and responsible person requirements
Canada: Health Canada regulations with distinct cosmetic and drug classifications
Asia-Pacific: Varying requirements across China, Japan, Korea, and Southeast Asian markets
Each market has different banned ingredients, labeling requirements, testing standards, and documentation needs. Navigating this alone feels impossible.
The Knowledge Gap
Most beauty founders are creatives, marketers, or product enthusiasts. They are not regulatory experts. Yet they are expected to understand complex chemical safety data, interpret evolving legislation, and maintain perfect documentation.
The Cost Fear
Many founders assume compliance requires massive budgets: expensive consultants, lengthy lab testing, and dedicated regulatory staff. For early-stage brands, these costs seem prohibitive.
The Real Cost of Non-Compliance
Before we discuss solutions, let us be clear about the stakes. Regulatory failures are not theoretical risks. They are business killers.
Real consequences include:
Product recalls that destroy brand reputation and customer trust
FDA warning letters that become public and damage credibility
Market access denial preventing sales in entire countries
Legal liability from consumer harm claims
Retail partnership termination when compliance issues surface
One violation can erase years of brand building. Prevention is always cheaper than cleanup.
How Smart Brands Eliminate Regulatory Anxiety

1. Start With Pre-Screened Formulations
The biggest compliance risk comes from ingredient selection. Many common cosmetic ingredients face restrictions or bans in major markets. Others require specific concentration limits or warning labels.
Smart approach: Use formulations that have already passed regulatory screening for your target markets.
The RhinoBird Solution: Our platform provides access to pre-vetted, market-ready formulas that have already undergone compliance review. Instead of guessing whether an ingredient is safe or legal, you start with formulations that meet FDA, EU, and global standards. This eliminates the primary source of regulatory risk from day one.
2. Implement Real-Time Compliance Screening
Traditional compliance happens late in the development process. You create a product, then send it for regulatory review. If issues surface, you face expensive reformulation or launch delays.
Smart approach: Build compliance into every stage of product development.
The RhinoBird Solution: Our smart compliance screening system checks your designs in real time. As you select ingredients, packaging, and labels, the system flags potential issues immediately. You get it right the first time instead of discovering problems after production.
This proactive approach saves months of delays and thousands in rework costs.
3. Automate Documentation and Tracking
Regulatory compliance requires extensive documentation: safety assessments, manufacturing records, ingredient sourcing, batch testing, and adverse event logs. Manual tracking is error-prone and time-consuming.
Smart approach: Use technology to automate documentation.
Key systems to implement:
Digital batch records with full traceability
Automated ingredient inventory with expiration tracking
Integrated adverse event reporting
Centralized regulatory document storage
When inspectors or retailers request documentation, you produce it instantly instead of scrambling through files.
4. Understand Market-Specific Requirements Before You Expand
Many brands create compliance problems by assuming what works in one market works everywhere. It does not.
Common expansion mistakes:
Using US-compliant formulas in the EU without reformulation
Ignoring China's animal testing requirements
Failing to appoint a Responsible Person for EU sales
Misclassifying cosmetics as drugs (or vice versa) in different jurisdictions
Smart approach: Map compliance requirements before entering any new market.
The RhinoBird Solution: We help brands navigate global market access by matching products with the right compliance pathways. Our system identifies which formulas need modification for specific markets and guides you through local registration requirements.
The Compliance Checklist for Beauty Brands
Use this checklist to audit your current compliance posture:
Table
If any area shows gaps, address them before your next product launch.
Common Compliance Mistakes to Avoid
❌ Assuming Natural Equals Safe
Natural ingredients face the same regulatory scrutiny as synthetics. Some natural substances are highly restricted or banned. Always verify regulatory status regardless of origin.
❌ Copying Competitor Labels
Just because a competitor uses certain language or ingredients does not mean it is compliant. They may operate under different regulations, or they may be non-compliant themselves.
❌ Ignoring Fragrance Regulations
"Fragrance" is not a free pass. All fragrance components must meet safety standards, and some markets require full fragrance disclosure.
❌ Making Drug Claims on Cosmetics
Claiming your moisturizer "treats acne" or your serum "reduces wrinkles" can trigger drug classification. This requires entirely different regulatory pathways. Keep cosmetic claims cosmetic.
❌ Skipping Stability Testing
Products that degrade or separate create safety risks and customer complaints. Stability testing is not optional; it is essential for determining shelf life and storage conditions.
Conclusion: Compliance Is Your Competitive Advantage
Regulatory compliance is not a burden. It is a business enabler. Brands that master compliance gain:
Faster market entry because products are ready for sale immediately
Retail confidence because buyers trust your documentation
Customer trust because your safety standards are visible and verifiable
Expansion readiness because you can enter new markets without starting from scratch
Risk protection because you avoid the disasters that destroy less prepared competitors
The brands that worry about compliance are the ones doing it manually. The brands that succeed build compliance into their systems from the start.
Stop worrying about regulatory risks. Start building systems that eliminate them.
Ready to launch with confidence? RhinoBird's smart compliance screening eliminates regulatory guesswork. From pre-vetted formulations to real-time design checks to global market access guidance, we help beauty brands launch safely and scale globally.
Launch Compliant Products with RhinoBird →
Frequently Asked Questions
Q: What is MoCRA and how does it affect my brand?
A: The Modernization of Cosmetics Regulation Act of 2022 is the biggest update to US cosmetic law in decades. It requires facility registration, product listing, serious adverse event reporting, and good manufacturing practices. All cosmetic brands selling in the US must comply.
Q: How much does compliance testing cost?
A: Basic safety testing ranges from $2,000 to $10,000 per product. Full regulatory consulting can cost $10,000 to $50,000. Platforms like RhinoBird reduce these costs by providing pre-tested formulations and automated screening.
Q: Can I sell in the EU with US-compliant products?
A: Not automatically. The EU has stricter ingredient restrictions and requires a safety assessment and Responsible Person. Some US formulas need modification for EU compliance.
Q: What happens if I ignore compliance?
A: At best, you face market access denial. At worst, FDA warning letters, product recalls, legal liability, and brand destruction. Compliance is not optional.
Q: How do I know if my ingredients are compliant?
A: Check FDA and EU banned/restricted ingredient lists. Better yet, use a platform with built-in ingredient screening that flags issues automatically.
About RhinoBird
RhinoBird is the one-stop cosmetics development platform helping beauty brands launch and scale with confidence. Our smart compliance screening, transparent pricing, and global market access ensure your products meet the highest regulatory standards from day one.